- Coronary Angiography/Cardiac Catheterisation
- Coronary stenting
- PCI in Acute Myocardial Infarction (Acute heart attack)
- PCI in Acute Coronary Syndrome (ACS)
- Coronary Thrombus Aspiration (thrombectomy)
- Complex transcatheter procedures for adult structural heart diseases (performed with a Heart Team)
- Complex transcatheter procedures for other heart diseases (in conjunction with Heart Team)
- Closure of patent foramen ovale
- Closure of atrial/ventricular septal defects
CORONARY ANGIOGRAPHY/CARDIAC CATHETERISATION
Coronary angiography is performed in an X-ray lab (known as the Catheterisation Lab). A small calibre plastic tube (known as catheter) is positioned non-surgically through the skin in the groin or the wrist into an artery (figure 1). The catheter is then moved without pain to the heart and contrast injection is delivered to the arteries that supply the heart muscles. This allows the cardiologist to delineate and study the coronary arteries in great details and to determine if there are significant narrowings or blockages of the arteries (figure 2, figure 3).
| Figure 1 |  |
| Figure 2. Coronary angiogram showing the right coronary artery (left panel) and the left coronary system (right panel) | 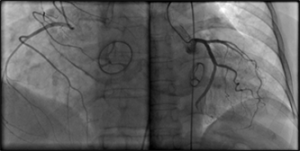 |
| Figure 3. Rotational angiography using specialised computerised program to see the right coronary artery in 180 degrees panning motion |
Cardiac catheterisation is performed in the same way as coronary angiography. The catheter is passed into the chambers of heart to find out detailed information about the structures of heart e.g. heart valves and measure pressures and oxygenation of the various heart chambers and large blood vessels of the heart. This procedure helps your cardiologist to precisely identify the heart problem(s) and make important decision(s) on the most appropriate cardiovascular therapy.
PERCUTANEOUS CORONARY INTERVENTION (PCI) AND CORONARY STENTING

Figure 1
This is a non-surgical method used to open narrowed arteries that supply heart muscles (coronary arteries). It is also known by a different variety of names – Percutaneous Transluminal Coronary Angioplasty (PTCA), Coronary Angioplasty. PCI is performed by inserting a catheter through the skin in the groin or arm into an artery. At the leading tip of this catheter, several different devices such as a balloon, stent, or cutting device (artherectomy device) can be deployed. The catheter and its devices are threaded through the inside of the artery into an area of coronary artery narrowing or blockage (see figure 1).
The first step involves the passing of a very fine non-traumatic wire (0.014” diameter) across the tight narrowed segment of the coronary artery followed by a balloon catheter monorailed to the site of narrowing and inflation of the balloon (with a diluted contrast fluid) to expand the narrowing by pushing the cholesterol plaques from within the lumen of the artery to the adjacent walls (see figure 2 and figure 3). Once a reasonable diameter of the lumen is achieved, a coronary stent (metallic scaffolding wire mesh) is deployed at the site of the narrowing to prop up the artery. The stent is designed to push back the cholesterol (fatty deposits) on the walls (i.e. “plastering”) and also functions to prevent elastic recoil of the coronary artery.

Figure 2
 Figure 3a. Pre-PCI/stent of the right coronary artery –circle showing at significant narrowing |  Figure 3b. Post PCI/stent deployment. The RCA (see circle) is now widely opened. |
What are coronary stents?
A coronary stent is a metallic wire mesh that functions as a scaffolding to support the coronary artery following balloon angioplasty to prevent elastic recoil. Unfortunately the presence of the metallic stent may result in tissue proliferation in part due to repair response of high pressure balloon inflation (barotrauma) and in part due to the presence of the foreign body metallic stent (leading to inflammatory response) with resultant re-narrowing of the artery.
Drug Eluting Stents (DES)
To prevent tissue proliferation with the metallic stent, anti-proliferative medications can be applied on the stent to inhibit the cells from proliferating (multiplying). The term “drug-eluting stent” (DES) is used for this type of stent. DES has shown to reduce re-narrowing of the coronary artery to single digit number (< 10%).
Bio absorbable Scaffoldings
Recently advancement in technology has allowed the use of plastic material to construct the stent which can dissolve in 2 to 3 years and allow the disappearance (dissolution) of the scaffoldings once the artery has healed and remodelled leaving no foreign body in the coronary artery.
TAVR
(TRANSCATHETER AORTIC VALVE REPLACEMENT)
Transcatheter Aortic Valve Implantation/ Replacement (TAVl/TAVR) is used to treat patients with Aortic Stenosis, a condition in which the aortic valve becomes narrowed, obstructing the outflow of blood from the heart and thereby requiring the heart to work harder to pump blood around the body.
Aortic Stenosis may lead to chest pain, shortness of breath, dizziness, loss of consciousness and even death. If left untreated, more than half of symptomatic patients do not live past two years. This is why treatment is so crucial for these patients.
The conventional treatment requires surgical repair and replacement of the valve through an open heart surgery. TAVR is a minimally invasive and non-surgical option to treat Aortic Stenosis. It is also designed for high-risk patients who are not suitable for conventional open heart surgery.
Typically, TAVR is recommended for patients who are more than 80 years of age or who are more than 65 years of age and have a very high risk for surgery. For these patients, certain criteria still need to be met and several tests are required to assess the suitability of the patient before proceeding further.
The procedure is performed in a special hybrid lab (a large operation theatre with the state-of-the-art imaging systems) by a Heart Team (comprising of interventional cardiologists, heart surgeon, echocardiologist, anaesthetist, theatre nurses, cardiac technicians and cardiac cath nurses, radiologist, radiographer, application specialists and an entire supporting team).
 Figure 1. Transcatheter aortic valve system in closed mode |  Figure 2. Transcatheter aortic valve in open mode |
 Figure 3. X-ray imaging of the transcatheter aortic valve |  Figure 4. Transcatheter aortic valve deployed in the heart |
TMVR
(TRANSCATHETER MITRAL VALVE REPAIR REPLACEMENT)
The MitraClip Therapy is used to treat Mitral Regurgitation (MR), a heart disorder in which the mitral valve does not close properly when the heart pumps out blood.
The mitral valve is located between the two chambers on the left side of the heart which directs blood flow in one direction – from the upper chamber (left atrium) to the lower chamber (left ventricle). When this valve does not close completely, MR or backflow of blood in the left ventricle occurs. In severe cases, reduced blood flow is pumped out of the heart. This creates excessive workload on the heart leading to dilation of the heart chambers. The condition can cause shortness of breath, fatigue, coughing, heart palpitations, swollen feet or ankles and heart murmur. If left untreated, MR’s progressive nature could lead to heart failure, stroke, heart attack or death.
There are currently several options of treatment available for MR including surgical treatment. The MitraClip Therapy is a less invasive catheter-based treatment option.
This procedure requires the same Heart Team as TAVR (see TAVR) and is also done in a specially-designed hybrid lab.